 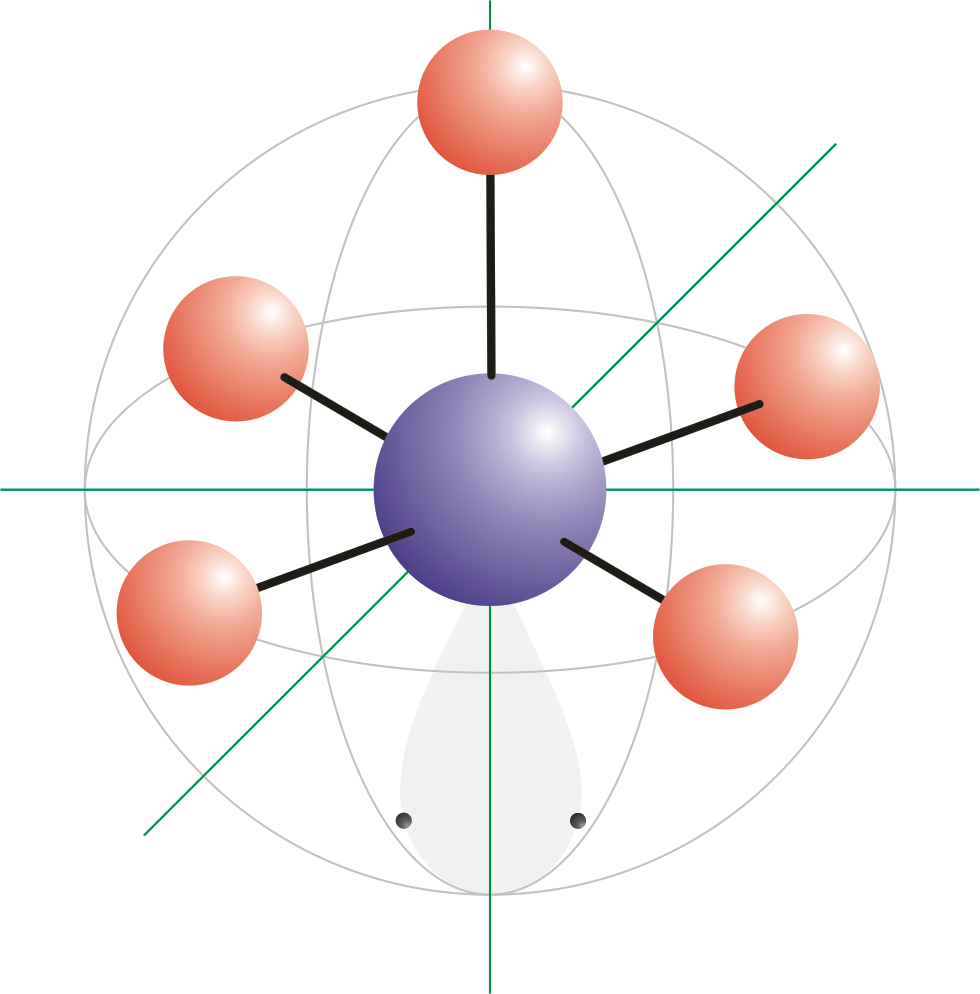
Thus, the molecule's shape reflects its equilibrium state in which it has the lowest possible energy in the system. The electrons and the nuclei settle into positions that minimize repulsion and maximize attraction. The shape of a molecule is determined by the location of the nuclei and its electrons. Using the VSEPR theory, the electron bond pairs and lone pairs on the center atom will help us predict the shape of a molecule. An electron group can be an electron pair, a lone pair, a single unpaired electron, a double bond or a triple bond on the center atom. VSEPR focuses not only on electron pairs, but it also focus on electron groups as a whole. Thus, electron pairs will spread themselves as far from each other as possible to minimize repulsion. The valence-shell electron-pair repulsion (VSEPR) theory states that electron pairs repel each other whether or not they are in bond pairs or in lone pairs. Now that we have a background in the Lewis electron dot structure we can use it to locate the the valence electrons of the center atom. Valence-Shell Electron-Pair Repulsion Theory This molecule has a square pyramidal shape because of the repelling forces set by the unbonded pairs of electrons of Fluorine and the additional unbonded pair of electrons from Iodine.\( \newcommand\) According to our structure we can say that the shape of our molecule is square pyramidal which derives from octahedral. The next step is to set up the Lewis Structure of the molecule. In this case the use of the chart is helpless as this molecule is an extended octet molecule. In the bottom or last row we conclude ny subtracting the second number from the first one and then divide that by 2 and we have the number of bonds. We add up all of the numbers from each column corresponding to their designated column. In the third column we write the number of electrons that we do have for each element. In the second column we write the number of electrons that are needed for the element to be “happy” or complete for each element. In the first column we write the elements we are using. 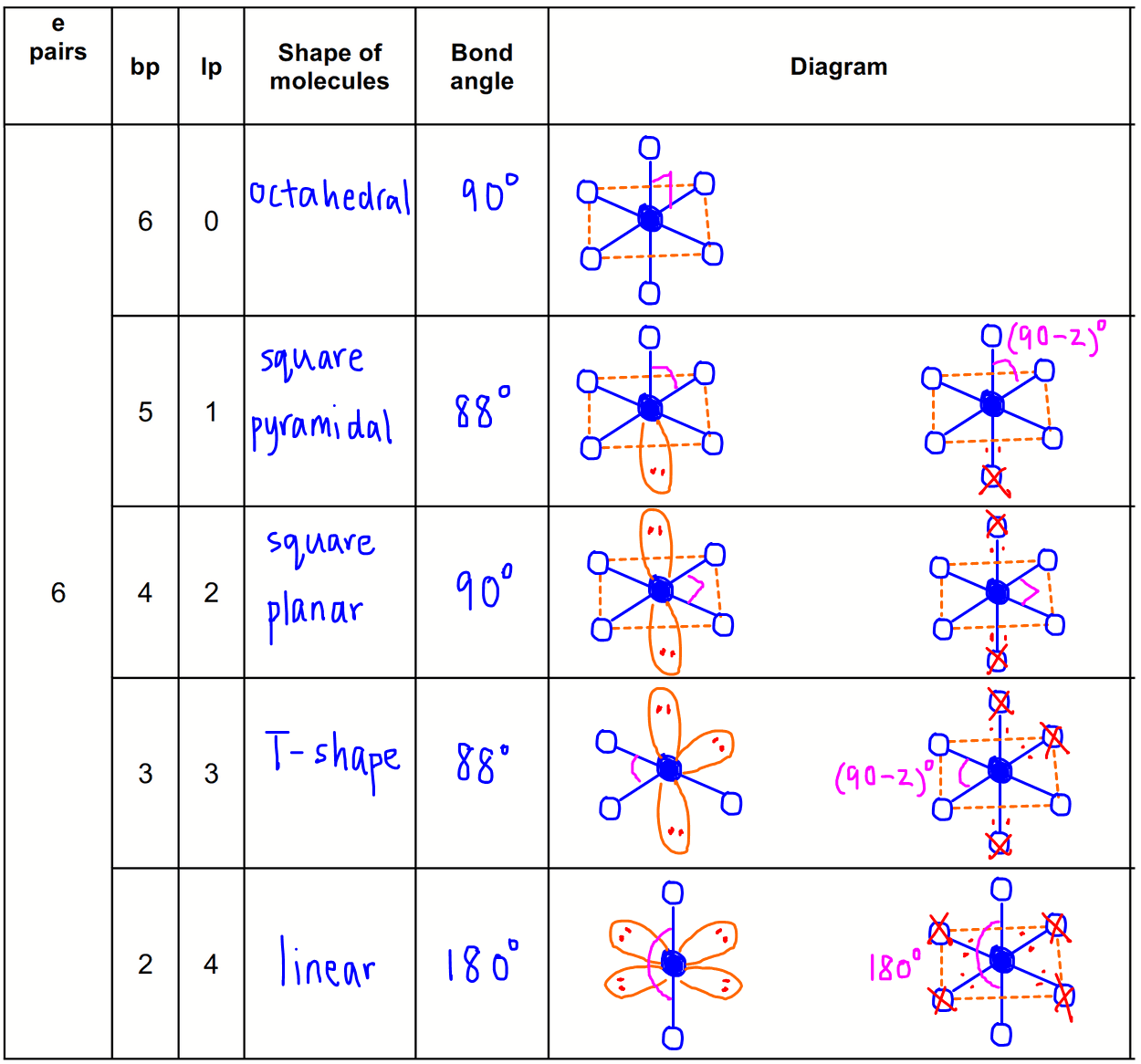
The chart is divided into 3 columns and 3 rows. If you want you may check the number of bonds that you are able to make with the application of the number of bonds chart. As we add up the number of electrons in bonds and alone we can conclude that this molecule does not satisfy the octet rule. Once we have formed the bonds we check for the octet rule or otherwise known as the rule of 8. 
However, since Iodine has 1 additional pair of unbonded electrons, the bonds formed with Fluorine are placed at angles of 90 and 120 degrees because of the repelling forces between them. Therefore, we can easily create 5 bonds, all the bonds surround the I, as I is our central atom. We already know that a bond between molecules is created by 2 electrons. For this problem I has 7 valence electrons and each F has 7 valence electrons. After we write the formula down we use a periodic table or our previous knowledge on the amount of valence electrons for the elements we use in order to set up the Lewis dot diagram. I’m now going to demonstrate how to bond Iodine Pentafluoride and what is its molecular geometry…Īs shown in the image above we begin this process by writing the formula of the molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
